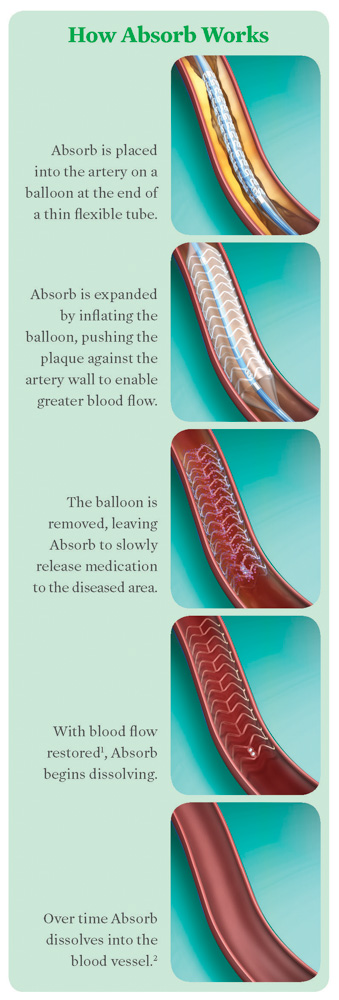
Coronary heart disease – damage or disease in the heart’s major blood vessels – affects approximately 15 million Americans and is the leading cause of death, worldwide. The usual cause is a build-up of plaque (fat deposits) on the inner walls of the coronary arteries, which narrows them and limits blood flow to the heart.
Genesys Heart Institute is now implanting a new, FDA-approved dissolvable stent in patients who suffer from coronary artery disease. Genesys is the first hospital in the region and one of only 50 in the entire country to offer the stent. Three other Ascension Michigan hospitals also offer this bioabsorbable scaffolding: St. Mary’s of Michigan-Saginaw, St. John Providence in the Detroit area, and Borgess Medical Center in Kalamazoo.
 This major advancement is a minimally-invasive procedure that is performed in the cardiac catheterization lab at Genesys. The catheter is inserted through a blood vessel in either the wrist or groin, and the device is used to open a clogged heart artery, restoring normal blood flow. After three to six months, the device will begin to gradually dissolve. After two to three years, it will dissolve completely and naturally once the vessel heals and can stay open on its own. With all of its steps – from preparing the vessel to completion – the procedure can take five to ten minutes, on average, which is longer than a standard stenting procedure.
This major advancement is a minimally-invasive procedure that is performed in the cardiac catheterization lab at Genesys. The catheter is inserted through a blood vessel in either the wrist or groin, and the device is used to open a clogged heart artery, restoring normal blood flow. After three to six months, the device will begin to gradually dissolve. After two to three years, it will dissolve completely and naturally once the vessel heals and can stay open on its own. With all of its steps – from preparing the vessel to completion – the procedure can take five to ten minutes, on average, which is longer than a standard stenting procedure.
“Bioabsorbable scaffolding” is the terminology preferred by interventional cardiologists and other industry leaders, as “stent” is now associated with the metal alloy materials that began emerging and became more ubiquitous since the early 1990s. Metal stents have traditionally been used in cath labs to open blocked arteries, but they are permanent. Once the artery is open, a stent serves no additional purpose; occasionally, it may contribute to future blockage.
“We have learned from basic research in animals and humans that once you fix the architecture of the vessel wall with this metal stent, the artery loses some of its biological function, such as adapting, expanding, and constricting,” says Dr. Abed Osman, Interventional Cardiologist at Genesys.
With bioabsorbable scaffolding, the artery can pulse and flex naturally as the heart demands for daily activities. This design provides the artery with the benefits of a traditional stent without some of the possible long-term concerns of a metal device. Interventional cardiologists exercise their best judgement for choosing either metal or bioabsorbable scaffolding for their patients. Before they decide to perform the procedure, a special intravascular imaging device may be used to determine the extent of blockage, and whether implementing a stent is the best option.
All interventional cardiologists at Genesys who can perform this procedure had to go “back to basics” in order to use the bioabsorbable scaffolding correctly, as deployment is quite different than the metal stent. This vigilance involves primarily learning which patient would likely benefit the most from this technology, selecting the right blockage as far as the size, extent, the length and type of artery, and the technique of deployment. In terms of the rate of deployment, the interventional cardiologists have to prepare the vessel, and then complete touch-ups and make sure it’s implanted well and fully expanded to perform its best function and reduce the risk of post-procedure complications.
Some complications include blockages re-growing inside and clots gathering more acutely.
To help reduce further complications, patients who receive either a metal or bioabsorbable stent must strictly adhere to prescription blood thinners for up to 12 months.
“It reminds us of the earlier days of the metal stent technology in the 1990s; we are a lot more meticulous about preparing the vessel,” Dr. Osman shares. “We use special intravascular imaging devices so the stenting result is successful. This field is going to evolve the same way as it did with metal stents. With the refinement of this technology, we expect to see improvement in engineering and design to the point where they still provide the same radial strength and at the same time make thinner struts, so patients will not face these challenges.”
Currently, researchers, engineers and clinical trials are working to make the bioabsorbable scaffolding’s radial strength stronger with thinner struts (a third-generation metal stent is 80-90 microns thick, as opposed to the bioabsorbable scaffolding at 140 microns).
“Right now, we are still being selective about who qualifies for this procedure,” Dr. Osman says. “But we have a very good product and many patients who would qualify for it. We are very excited to offer this to our patients here at Genesys. We have a vibrant research department and enthusiastic group of physicians and health care professionals who are willing to provide the best and latest technology for our patients.”
Photography by Eric Dutro and graphics provided by Genesys/Absorb
Genesys is the first hospital in the region and one of only 50 in the country to offer the dissolvable stent.